Etiquetado: cetoacidosis

Cetoacidosis diabética
ketosis is the body’s natural adaptation to starvation and is not to be confused with the dangerous ketoacidosis associated with untreated type 1 diabetes (fuente)
la cetosis es la adaptación natural del cuerpo a la inanición y no hay que confundirla con la peligrosa cetoacidosis asociada a la diabetes tipo 1 no tratada
Cetoacidosis
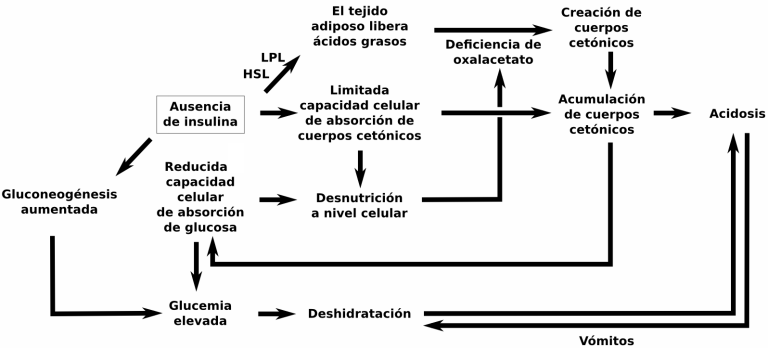
(NOTA: el esquema es de elaboración propia. Puede no ser correcto)
Algunas citas:
Ketones only rise to dangerous levels in people who have type I diabetes and can’t make their own insulin (Michael R. Eades)
DIABETIC KETOACIDOSIS AND THE HYPERGLYCEMIC hyperosmolar state are the most serious complications of diabetic decompensation and remain associated with excess mortality. Insulin deficiency is the main underlying abnormality (fuente)
during very low carbohydrate intake, the regulated and controlled production of ketone bodies causes a harmless physiological state known as dietary ketosis. In ketosis, the blood pH remains buffered within normal limits (fuente)
Thus glucose transport and uptake into cells are dependent on the degree of ketosis. In the face of extreme ketosis, such as that not uncommonly seen in type 1 diabetes, where concentrations rise to as high as 25 mmol litre–1, glucose uptake can be reduced to almost zero in tissues, such as muscle, that can metabolize ketones as readily as glucose (fuente)
In DKA, the deficiency of insulin activates lipolysis in adipose tissue releasing increased FFA, which accelerates formation of VLDL in the liver. In addition, reduced activity of lipoprotein lipase in peripheral tissue decreases removal of VLDL from the plasma, resulting in hypertriglyceridemia (fuente)
Even under conditions of extreme ketoacidosis there is no significant membrane barrier to glucose uptake – the block occurs «lower down» in the metabolic pathway where the excess of ketones competitively blocks the metabolites of glucose entering the citric acid cycle (fuente)
To be utilized as an energy source, FFAs must be transported from the peripheral tissues into the mitochondria of hepatocytes. Once inside the mitochondria, FFAs undergo beta oxidation which converts them into acetyl coenzyme A (acetyl-CoA). Under normal circumstances acetyl-CoA enters the tricarboxylic cycle. To do this, acetyl-CoA first needs to pair with oxaloacetate which is derived from pyruvate during glycolysis. In states of decreased intracellular glucose concentration such as DKA, oxaloacetate will be deficient as it will be preferentially shifted into the gluconeogenesis pathway. The oxaloacetate deficiency, when combined with overproduction of acetyl-CoA, will shift the further metabolism of acetyl-CoA towards ketone body formation […] Limited tissue uptake capacity of ketone bodies is primarily mediated by the effective lack of insulin. Overproduction of ketones rather than decreased utilization, however, appears to be the primary mechanism of ketone body accumulation during DKA (fuente)
In order to satisfy its cellular energy requirements and maintain cellular integrity, the body utilizes adipose tissue as the main energy source (1,4). […] Multiple metabolic pathways are involved in this shift in energy utilization. Hormone sensitive lipase, the activity of which is normally inhibited by insulin, is the main mediator of this process (1–4). This enzyme mediates the degradation of triglycerides and formation of free fatty acids (FFA) (fuente)
Liver fatty acid β oxidation is high in the fasted state and low in the fed state. Mitochondrial β oxidation not only provides energy for hepatocytes but also generates ketone bodies (β-hydroxybutyrate, acetoacetate, and acetone) which are exported into the circulation and provide metabolic fuels for extrahepatic tissues during fasting.[…] In the fed state, insulin stimulates phosphorylation of PGC-1α by Akt, which impairs the ability of PGC-1α to stimulate fatty acid β oxidation (fuente)
Leer más:
- ¿Existe diferencia entre cetosis y cetoacidosis? por Ana Muñiz